NEET 2025 — Code 48
Official NTA NEET-UG 2025 question paper, set 48. 180 questions across Physics, Chemistry, and Biology with worked solutions and the official answer key.
A parallel plate capacitor made of circular plates is being charged such that the surface charge density on its plates is increasing at a constant rate with time. The magnetic field arising due to displacement current is:
Non-zero everywhere with maximum at the imaginary cylindrical surface connecting peripheries of the plates
Zero between the plates and non-zero outside
Zero at all places
Constant between the plates and zero outside the plates
An electric dipole with dipole moment is aligned with the direction of a uniform electric field of magnitude . The dipole is then rotated through an angle of with respect to the electric field. The change in the potential energy of the dipole is:
2 J
5 J
0.8 J
0 J
A ball of mass 0.5 kg is dropped from a height of 40 m. The ball hits the ground and rises to a height of 10 m. The impulse imparted to the ball during its collision with the ground is (Take )
0
84 NS
21 NS
7 NS
The intensity of transmitted light when a polaroid sheet, placed between two crossed polaroids at from the polarization axis of one of the polaroids, is ( is the intensity of polarised light after passing through the first polaroid):
The kinetic energies of two similar cars A and B are 100 J and 225 J respectively. On applying brakes, car A stops after 1000 m and car B stops after 1500 m. If and are the forces applied by the brakes on cars A and B respectively, then the ratio of is
The current passing through the battery in the given circuit, is:
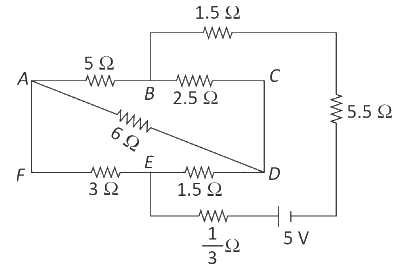
0.5 A
1.5 A
2.0 A
2.5 A
Three identical heat conducting rods are connected in series as shown in the figure. The rods on the sides have thermal conductivity while that in the middle has thermal conductivity . The left end of the combination is maintained at temperature and the right end at . The rods are thermally insulated from outside. In steady state, temperature at the left junction is and that at the right junction is . The ratio is
In an oscillating spring mass system, a spring is connected to a box filled with sand. As the box oscillates, sand leaks slowly out of the box vertically so that the average frequency and average amplitude of the system change with time . Which one of the following options schematically depicts these changes correctly?
AB is a part of an electrical circuit (see figure). The potential difference "", at the instant when current and is increasing at a rate of 1 amp/second is:
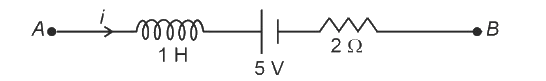
9 volt
10 volt
5 volt
6 volt
A particle of mass is moving around the origin with a constant force pulling it towards the origin. If Bohr model is used to describe its motion, the radius of the orbit and the particle's speed in the orbit depend on as
In some appropriate units, time (t) and position (x) relation of a moving particle is given by . The acceleration of the particle is
A model for quantized motion of an electron in a uniform magnetic field states that the flux passing through the orbit of the electron is where is an integer, is Planck's constant and is the magnitude of electron's charge. According to the model, the magnetic moment of an electron in its lowest energy state will be ( is the mass of the electron)
A microscope has an objective of focal length 2 cm, eyepiece of focal length 4 cm and the tube length of 40 cm. If the distance of distinct vision of eye is 25 cm, the magnification in the microscope is
150
250
100
125
There are two inclined surfaces of equal length () and same angle of inclination with the horizontal. One of them is rough and the other is perfectly smooth. A given body takes 2 times as much time to slide down on rough surface than on the smooth surface. The coefficient of kinetic friction () between the object and the rough surface is close to
0.5
0.75
0.25
0.40
A full wave rectifier circuit with diodes () and () is shown in the figure. If input supply voltage volt, then at msec
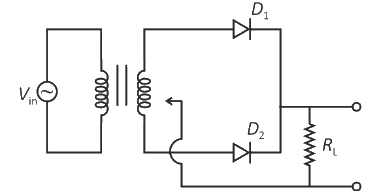
and both are forward biased
and both are reverse biased
is forward biased, is reverse biased
is reverse biased, is forward biased.
A uniform rod of mass 20 kg and length 5 m leans against a smooth vertical wall making an angle of with it. The other end rests on a rough horizontal floor. The friction force that the floor exerts on the rod is (Take )
200 N
100 N
Two identical charged conducting spheres A and B have their centres separated by a certain distance. Charge on each sphere is and the force of repulsion between them is . A third identical uncharged conducting sphere is brought in contact with sphere A first and then with B and finally removed from both. New force of repulsion between spheres A and B (Radii of A and B are negligible compared to the distance of separation so that for calculating force between them they can be considered as point charges) is best given as:
Two cities X and Y are connected by a regular bus service with a bus leaving in either direction every min. A girl is driving scooty with a speed of 60 km/h in the direction X to Y notices that a bus goes past her every 30 minutes in the direction of her motion, and every 10 minutes in the opposite direction. Choose the correct option for the period of the bus service and the speed (assumed constant) of the buses.
10 min, 90 km/h
15 min, 120 km/h
9 min, 40 km/h
25 min, 100 km/h
A container has two chambers of volumes litres and litres separated by a partition made of a thermal insulator. The chambers contain and moles of ideal gas at pressures atm and atm, respectively. When the partition is removed, the mixture attains an equilibrium pressure of
4 atm
8 atm
3 atm
6 atm
De-Broglie wavelength of an electron orbiting in the state of hydrogen atom is close to (Given Bohr radius = 0.052 nm)
67 nm
67 nm
0.067 nm
0.67 nm
To an ac power supply of 220 V at 50 Hz, a resistor of 20 , a capacitor of reactance 25 and an inductor of reactance 45 are connected in series. The corresponding current in the circuit and the phase angle between the current and the voltage is, respectively
15.6 A and 30°
15.6 A and 45°
7.8 A and 30°
7.8 A and 45°
Which of the following options represent the variation of photoelectric current with property of light shown on the x-axis?
A pipe open at both ends has a fundamental frequency in air. The pipe is now dipped vertically in a water drum to half of its length. The fundamental frequency of the air column is now equal to:
Two identical point masses and , suspended from two separate massless springs of spring constants and , respectively, oscillate vertically. If their maximum speeds are the same, the ratio of the amplitude of mass to the amplitude of mass is
The output (Y) of the given logic implementation is similar to the output of an/a ____ gate.
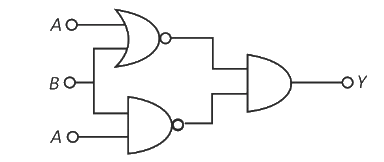
OR
NOR
AND
NAND
An oxygen cylinder of volume 30 litre has 18.20 moles of oxygen. After some oxygen is withdrawn from the cylinder, its gauge pressure drops to 11 atmospheric pressure at temperature 27°C. The mass of the oxygen withdrawn from the cylinder is nearly equal to: [Given, , and molecular mass of , 1 atm pressure = ]
0.116 kg
0.156 kg
0.125 kg
0.144 kg
In a certain camera, a combination of four similar thin convex lenses are arranged axially in contact. Then the power of the combination and the total magnification in comparison to the power () and magnification () for each lens will be, respectively
and
and
and
and
Two gases A and B are filled at the same pressure in separate cylinders with movable pistons of radius and , respectively. On supplying an equal amount of heat to both the systems reversibly under constant pressure, the pistons of gas A and B are displaced by 16 cm and 9 cm, respectively. If the change in their internal energy is the same, then the ratio is equal to
A balloon is made of a material of surface tension and its inflation outlet (from where gas is filled in it) has small area . It is filled with a gas of density and takes a spherical shape of radius . When the gas is allowed to flow freely out of it, its radius changes from to (zero) in time . If the speed of gas coming out of the balloon depends on as and then
A bob of heavy mass is suspended by a light string of length . The bob is given a horizontal velocity as shown in figure. If the string gets slack at some point making an angle from the horizontal, the ratio of the speed of the bob at point to its initial speed is:
A physical quantity is related to four observations and as follows: The percentage errors of measurement in and are , , , and respectively. The percentage error in the quantity is
13%
15%
10%
2%
The Sun rotates around its centre once in 27 days. What will be the period of revolution if the Sun were to expand to twice its present radius without any external influence? Assume the Sun to be a sphere of uniform density.
115 days
108 days
100 days
105 days
The radius of Martian orbit around the Sun is about 4 times the radius of the orbit of Mercury. The Martian year is 687 Earth days. Then which of the following is the length of 1 year on Mercury?
172 earth days
124 earth days
88 earth days
225 earth days
A wire of resistance is cut into 8 equal pieces. From these pieces two equivalent resistances are made by adding four of these together in parallel. Then these two sets are added in series. The net effective resistance of the combination is:
A photon and an electron (mass ) have the same energy . The ratio () of their de Broglie wavelengths is: ( is the speed of light)
A sphere of radius is cut from a larger solid sphere of radius as shown in the figure. The ratio of the moment of inertia of the smaller sphere to that of the rest part of the sphere about the Y-axis is:
1:1
1:4
1:2
1:3
An electron (mass kg and charge C) moving with speed ( speed of light) is injected into a magnetic field of magnitude T perpendicular to its direction of motion. We wish to apply an uniform electric field together with the magnetic field so that the electron does not deflect from its path. Then (speed of light ms)
is parallel to and its magnitude is V m
is parallel to and its magnitude is V m
is perpendicular to and its magnitude is V m
is perpendicular to and its magnitude is V m
The electric field in a plane electromagnetic wave is given by . Then expression for the corresponding magnetic field is (here subscripts denote the direction of the field):
A body weighs 48 N on the surface of the earth. The gravitational force experienced by the body due to the earth at a height equal to one-third the radius of the earth from its surface is :
32 N
36 N
16 N
27 N
An unpolarized light beam travelling in air is incident on a medium of refractive index 1.73 at Brewster’s angle. Then
Both reflected and transmitted light are perfectly polarized with angles of reflection and refraction close to 60° and 30°, respectively
Transmitted light is completely polarized with angle of refraction close to 30°
Reflected light is completely polarized and the angle of reflection is close to 60°
Reflected light is partially polarized and the angle of reflection is close to 30°
A constant voltage of 50 V is maintained between the points A and B of the circuit shown in the figure. The current through the branch CD of the circuit is:
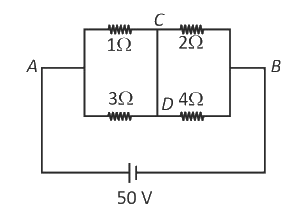
5 A
0 A
5 A
0 A
The plates of a parallel plate capacitor are separated by . Two slabs of different dielectric constant and with thickness and , respectively are inserted in the capacitor. Due to this, the capacitance becomes two times larger than when there is nothing between the plates. If , the value of is:
60
33
66
33
Consider the diameter of a spherical object being measured with the help of a Vernier callipers. Suppose its 10 Vernier Scale Divisions (V.S.D.) are equal to its 9 Main Scale Divisions (M.S.D.). The least division in the M.S. is 0.1 cm and the zero of V.S. is at cm when the jaws of Vernier callipers are closed. If the main scale reading for the diameter is cm and the number of coinciding vernier division is 8, the measured diameter after zero error correction, is
98 cm
5.00 cm
5.18 cm
5.08 cm
A 2 amp current is flowing through two different small circular copper coils having radii ratio 1 : 2. The ratio of their respective magnetic moments will be
2 : 1
4 : 1
1 : 4
1 : 2
Consider a water tank shown in the figure. It has one wall at and can be taken to be very wide in the direction. When filled with a liquid of surface tension and density , the liquid surface makes angle () with the -axis at . If is the height of the surface then the equation for is: (take , is the acceleration due to gravity)
Identify the suitable reagent for the following conversion.
(i) , (ii)
(i) , (ii)
(i) , (ii)
The correct order of decreasing acidity of the following aliphatic acids is
HCOOH > CH₃COOH > (CH₃)₂CHCOOH > (CH₃)₃CCOOH
HCOOH > (CH₃)₃CCOOH > (CH₃)₂CHCOOH > CH₃COOH
(CH₃)₃CCOOH > (CH₃)₂CHCOOH > CH₃COOH > HCOOH
CH₃COOH > (CH₃)₂CHCOOH > (CH₃)₃CCOOH > HCOOH
Which one of the following reactions does NOT belong to "Lassaigne's test"?
If the rate constant of a reaction is , how much time does it take for concentration of the reactant to get reduced to ? (Given: )
Given below are two statements : Statement I : A hypothetical diatomic molecule with bond order zero is quite stable. Statement II : As bond order increases, the bond length increases. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is true but Statement II is false
Statement I is false but Statement II is true
Both Statement I and Statement II are true
Both Statement I and Statement II are false
Out of the following complex compounds, which of the compound will be having the minimum conductance in solution?
[Co(NH)Cl]
[Co(NH)Cl]Cl
[Co(NH)Cl]
[Co(NH)]Cl
Which of the following aqueous solution will exhibit highest boiling point?
0.01M Na₂SO₄
0.015M C₆H₁₂O₆
0.01M Urea
0.01M KNO₃
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A): Compound (I) undergoes reaction faster than Compound (II). Reason (R): Iodine is a better leaving group because of its large size. In the light of the above statements, choose the correct answer from the options given below:
A is true but R is false
A is false but R is true
Both A and R are true and R is the correct explanation of A
Both A and R are true but R is not the correct explanation of A
Consider the following compounds : , and The oxidation state of the underlined elements in them are, respectively,
+1, -2, and +4
+4, -4, and +6
+1, -1, and +6
+2, -2, and +6
Match List-I with List-II.
A-I, B-II, C-III, D-IV
A-I, B-IV, C-III, D-II
A-I, B-II, C-IV, D-III
A-II, B-III, C-I, D-IV
Given below are two statements : Statement I : Like nitrogen that can form ammonia, arsenic can form arsine. Statement II : Antimony cannot form antimony pentoxide. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but Statement II is incorrect
Statement I is incorrect but Statement II is correct
Both Statement I and Statement II are correct
Both Statement I and Statement II are incorrect
Given below are two statements : Statement I : Ferromagnetism is considered as an extreme form of paramagnetism. Statement II : The number of unpaired electrons in a Cr²⁺ ion (Z = 24) is the same as that of a Nd³⁺ ion (Z = 60). In the light of the above statements, choose the correct answer from the options given below :
Statement I is true but Statement II is false
Statement I is false but Statement II is true
Both Statement I and Statement II are true
Both Statement I and Statement II are false
Which one of the following reactions does NOT belong to "Lassaigne's test"?
Match List-I with List-II.
A-I, B-II, C-III, D-IV
A-I, B-II, C-IV, D-III
A-II, B-III, C-I, D-IV
A-I, B-IV, C-III, D-II
How many products (including stereoisomers) are expected from monochlorination of the following compound?
5
6
2
3
Which of the following statements are true? A. Unlike Ga that has a very high melting point, Cs has a very low melting point. B. On Pauling scale, the electronegativity values of N and Cl are not the same. C. Ar, K⁺, Cl⁻, Ca²⁺, and S²⁻ are all isoelectronic species. D. The correct order of the first ionization enthalpies of Na, Mg, Al, and Si is Si > Al > Mg > Na. E. The atomic radius of Cs is greater than that of Li and Rb. Choose the correct answer from the options given below :
C and D only
A, C, and E only
A, B, and E only
C and E only
The standard heat of formation, in kcal/mol of is : [Given : standard heat of formation of ion (aq) = kcal/mol, standard heat of crystallisation of = kcal/mol, standard heat of formation of = kcal/mol]
Match List-I with List-II
| List-I (Example) | List-II (Type of Solution) |
| A. Humidity | I. Liquid in gas |
| B. Alloys | II. Solid in solid |
| C. Amalgams | IV. Liquid in solid |
| D. Smoke | III. Solid in gas |
A-III, B-I, C-IV, D-II
A-III, B-II, C-I, D-IV
A-II, B-IV, C-I, D-III
A-II, B-I, C-IV, D-III
C(s) + 2H₂(g) → CH₄(g); kJ mol⁻¹. Which of the following diagrams gives an accurate representation of the above reaction? [R → reactants; P → products]
Sugar 'X' A. is found in honey B. is a keto sugar C. exists in and - anomeriс forms. D. Is laevorotatory. 'X' is :
Maltose
Sucrose
D-Glucose
D-Fructose
Total number of possible isomers (both structural as well as stereoisomers) of cyclic ethers of molecular formula is :
10
11
6
8
For the reaction , the backward reaction rate constant is higher than the forward reaction rate constant by a factor of 2500, at 1000 K. [Given : ] for the reaction at 1000 K is
0.033
0.021
83.1
The ratio of the wavelengths of the light absorbed by a Hydrogen atom when it undergoes and transitions, respectively, is
If the molar conductivity () of a 0.050 mol L solution of a monobasic weak acid is 90 S cm mol, its extent (degree) of dissociation will be [Assume S cm mol and S cm mol.]
0.225
0.215
0.115
0.125
5 moles of liquid X and 10 moles of liquid Y make a solution having a vapour pressure of 70 torr. The vapour pressures of pure X and Y are 63 torr and 78 torr respectively. Which of the following is true regarding the described solution?
The solution is ideal.
The solution has volume greater than the sum of individual volumes.
The solution shows positive deviation.
The solution shows negative deviation.
Among the following, choose the ones with equal number of atoms. A. 212 g of [molar mass = 106 g] B. 248 g of [molar mass = 62 g] C. 240 g of [molar mass = 40 g] D. 12 g of [molar mass = 2 g] E. 220 g of [molar mass = 44 g]
B, C, and D only
B, D, and E only
A, B, and C only
A, B, and D only
Which of the following are paramagnetic?
[NiCl₄]²⁻
Ni(CO)₄
[Ni(CN)₄]²⁻
[Ni(H₂O)₆]²⁺
If the half-life (t₁/₂) for a first order reaction is 1 minute, then the time required for 99.9% completion of the reaction is closest to :
5 minutes
10 minutes
2 minutes
4 minutes
Energy and radius of first Bohr orbit of He⁺ and Li²⁺ are [Given R_H = 2.18 × 10⁻¹⁸ J, a₀ = 52.9 pm]
E_n(Li²⁺) = -19.62 × 10⁻¹⁸ J; r_n(Li²⁺) = 17.6 pm E_n(He⁺) = -8.72 × 10⁻¹⁸ J; r_n(He⁺) = 26.4 pm
E_n(Li²⁺) = -8.72 × 10⁻¹⁶ J; r_n(Li²⁺) = 17.6 pm E_n(He⁺) = -19.62 × 10⁻¹⁶ J; r_n(He⁺) = 17.6 pm
E_n(Li²⁺) = -19.62 × 10⁻¹⁶ J; r_n(Li²⁺) = 17.6 pm E_n(He⁺) = -8.72 × 10⁻¹⁶ J; r_n(He⁺) = 26.4 pm
E_n(Li²⁺) = -8.72 × 10⁻¹⁸ J; r_n(Li²⁺) = 26.4 pm E_n(He⁺) = -19.62 × 10⁻¹⁸ J; r_n(He⁺) = 17.6 pm
Among the given compounds I-III, the correct order of bond dissociation energy of C–H bond marked with * is :
II > I > III
I > II > III
III > II > I
II > III > I
Dalton's Atomic theory could not explain which of the following?
Law of multiple proportion
Law of gaseous volume
Law of conservation of mass
Law of constant proportion
Identify the correct orders against the property mentioned A. – dipole moment B. – number of lone pairs on central atom C. – bond length D. – bond enthalpy
A, C only
B, C only
A, D only
B, D only
Match List-I with List-II.
| List-I (Name of Vitamin) | List-II (Deficiency disease) |
| A. Vitamin B₁₂ | I. Pernicious anaemia |
| B. Vitamin D | II. Rickets |
| C. Vitamin B₂ | III. Cheilosis |
| D. Vitamin B₆ | IV. Convulsions |
A-II, B-III, C-I, D-IV
A-IV, B-III, C-II, D-I
A-I, B-II, C-II, D-IV
A-IV, B-III, C-I, D-II
The correct order of decreasing basic strength of the given amines is:
N-ethylethanamine > ethanamine > N-methylaniline > benzenamine
benzenamine > ethanamine > N-methylaniline > N-ethylethanamine
N-methylaniline > benzenamine > ethanamine > N-ethylethanamine
N-ethylethanamine > ethanamine > benzenamine > N-methylaniline
The correct order of the wavelength of light absorbed by the following complexes is, A. B. C. D. Choose the correct answer from the options given below:
Which one of the following compounds does not decolourize bromine water?
Predict the major product P in the following sequence of reactions:
Match List-I with List-II.
A-II, B-III, C-I, D-IV
A-I, B-II, C-III, D-IV
A-I, B-IV, C-III, D-II
A-I, B-II, C-IV, D-III
Which among the following electronic configurations belong to main group elements? A. B. C. D. E. Choose the correct answer from the option given below :
B and E only
A and C only
D and E only
A, C and D only
Which one of the following compounds can exist as cis-trans isomers?
1,1-Dimethylcyclopropane
1,2-Dimethylcyclohexane
Pent-1-ene
2-Methylhex-2-ene
Phosphoric acid ionizes in three steps with their ionization constant values , and , respectively, while is the overall ionization constant. Which of the following statements are true? A. B. is a stronger acid than and C. D.
B, C and D only
A, B and C only
A and B only
A and C only
Match List I with List II
| List-I (Ion) | List-II (Group Number in Cation Analysis) |
| A. Co²⁺ | I. Group-I |
| B. Mg²⁺ | II. Group-III |
| C. Pb²⁺ | III. Group-IV |
| D. Al³⁺ | IV. Group-VI |
A-III, B-II, C-IV, D-I
A-III, B-II, C-I, D-IV
A-III, B-IV, C-II, D-I
A-III, B-IV, C-I, D-II
Higher yield of NO in can be obtained at [ of the reaction = +180.7 kJ mol⁻¹]
Higher temperature
Lower temperature
Higher concentration of N₂
Higher concentration of O₂
Given below are two statements : Statement-I : Benzenediazonium salt is prepared by the reaction of aniline with nitrous acid at 273 – 278 K. It decomposes easily in the dry state. Statement-II : Insertion of iodine into the benzene ring is difficult and hence iodobenzene is prepared through the reaction of benzenediazonium salt with KI. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but Statement II is incorrect
Statement I is incorrect but Statement II is correct
Both Statement I and Statement II are correct
Both Statement I and Statement II are incorrect
Predict the major product P in the following sequence of reactions:
In the above represented plasmid, an alien piece of DNA is inserted at EcoRI site. Which of the following strategies will be chosen to select the recombinant colonies?
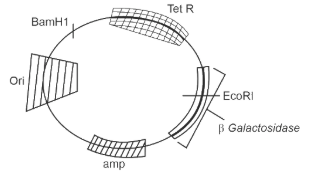
White color colonies will be selected.
Blue color colonies grown on ampicillin plates can be selected.
Using ampicillin & tetracycline containing medium plate.
Blue color colonies will be selected.
The protein portion of an enzyme is called:
Apoenzyme
Prosthetic group
Cofactor
Coenzyme
Given below are two statements: Statement I : The primary source of energy in an ecosystem is solar energy. Statement II : The rate of production of organic matter during photosynthesis in an ecosystem is called net primary productivity (NPP). In the light of the above statements, choose the most appropriate answer from the options given below:
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
Given below are two statements : One is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : A typical unfertilised, angiosperm embryo sac at maturity is 8 nucleate and 7-celled. Reason (R) : The egg apparatus has 2 polar nuclei. In the light of the above statements, choose the correct answer from the options given below :
A is true but R is false
A is false but R is true
Both A and R are true and R is the correct explanation of A
Both A and R are true but R is NOT the correct explanation of A
Neoplastic characteristics of cells refer to : A. A mass of proliferating cell B. Rapid growth of cells C. Invasion and damage to the surrounding tissue D. Those confined to original location Choose the correct answer from the options given below:
A, B, D only
B, C, D only
A, B only
A, B, C only
Which one of the following is the characteristic feature of gymnosperms?
Seeds are absent
Gymnosperms have flowers for reproduction
Seeds are enclosed in fruits
Seeds are naked
Match List-I with List-II.
| List-I | List-II |
| A. Progesterone | I. Pars intermedia |
| B. Relaxin | II. Ovary |
| C. Melanocyte stimulating hormone | III. Adrenal Medulla |
| D. Catecholamines | IV. Corpus luteum |
A-II, B-IV, C-I, D-III
A-III, B-II, C-IV, D-I
A-IV, B-II, C-I, D-III
A-IV, B-II, C-III, D-I
Which chromosome in the human genome has the highest number of genes?
Chromosome 1
Chromosome 10
Chromosome X
Chromosome Y
Which of the following statements about RuBisCO is true?
It is an enzyme involved in the photolysis of water
It catalyzes the carboxylation of RuBP
It is active only in the dark
It has higher affinity for oxygen than carbon dioxide
The first menstruation is called :
Diapause
Ovulation
Menopause
Menarche
Which of the following genetically engineered organisms was used by Eli Lilly to prepare human insulin?
Virus
Phage
Bacterium
Yeast
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : All vertebrates are chordates but all chordates are not vertebrate. Reason (R) : The members of subphylum vertebrata possess notochord during the embryonic period, the notochord is replaced by cartilaginous or bony vertebral column in adults. In the light of the above statements, choose the correct answer from the options given below:
(A) is true but (R) is false
(A) is false but (R) is true
Both (A) and (R) are true and (R) is the correct explanation of (A)
Both (A) and (R) are true but (R) is not the correct explanation of (A)
What is the main function of the spindle fibers during mitosis?
To repair damaged DNA
To regulate cell growth
To separate the chromosomes
To synthesize new DNA
Match List I with List II:
| List-I | List-II | |
|---|---|---|
| A | Alfred Hershey and Martha Chase | I. DNA as genetic material confirmation |
| B | Euchromatin | III. Loosely packed and light-stained |
| C | Frederick Griffith | I. Streptococcus pneumoniae |
| D | Heterochromatin | II. Densely packed and dark-stained |
A-IV, B-III, C-I, D-II
A-III, B-II, C-IV, D-I
A-II, B-IV, C-I, D-III
A-IV, B-II, C-I, D-III
Match List I with List II:
| List-I | List-II |
| A. Adenosine | III. Nucleoside |
| B. Adenylic acid | II. Nucleotide |
| C. Adenine | I. Nitrogen base |
| D. Alanine | IV. Amino acid |
A-III, B-II, C-I, D-IV
A-II, B-III, C-I, D-IV
A-III, B-IV, C-II, D-I
A-III, B-II, C-IV, D-I
In frog, the Renal portal system is a special venous connection that acts to link :
Kidney and intestine
Kidney and lower part of body
Liver and intestine
Liver and kidney
Which of the following are the post-transcriptional events in an eukaryotic cell?
Transport of pre-mRNA to cytoplasm prior to splicing.
Removal of introns and joining of exons.
Addition of methyl group at 5′ end of hnRNA.
Addition of adenine residues at 3′ end of hnRNA.
Polymerase chain reaction (PCR) amplifies DNA following the equation.
Given below are two statements: One is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A): Both wind and water pollinated flowers are not very colourful and do not produce nectar. Reason (R): The flowers produce enormous amount of pollen grains in wind and water pollinated flowers. In the light of the above statements, choose the correct answer from the options given below:
A is true but R is false
A is false but R is true
Both A and R are true and R is the correct explanation of A
Both A and R are true but R is NOT the correct explanation of A
Epiphytes that are growing on a mango branch is an example of which of the following?
Predation
Amensalism
Commensalism
Mutualism
Find the correct statement : (A) In human pregnancy, the major organ systems are formed at the end of 12 weeks. (B) In human pregnancy the major organ systems are formed at the end of 8 weeks. (C) In human pregnancy heart is formed after one month of gestation. (D) In human pregnancy, limbs and digits develop by the end of second month. (E) In human pregnancy the appearance of hair is usually observed in the fifth month. Choose the correct answer from the options given below :
B, C, D and E only
A, C, D and E only
A and E only
B and C only
Identify the part of a bio-reactor which is used as a foam braker from the given figure.
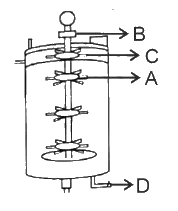
D
C
A
B
Frogs respire in water by skin and buccal cavity and on land by skin, buccal cavity and lungs. Choose the correct answer from the following :
The statement is false for water but true for land
The statement is false for both the environment
The statement is true for water but false for land
The statement is true for both the environment
Consider the following statements regarding function of adrenal medullary hormones : (A) It causes pupillary constriction. (B) It is a hyperglycemic hormone. (C) It causes piloerection. (D) It increases strength of heart contraction. Choose the correct answer from the options given below :
A, C and D only
D only
C and D only
B, C and D only
Read the following statements on plant growth and development. (A) Parthenocarpy can be induced by auxins. (B) Plant growth regulators can be involved in promotion as well as inhibition of growth. (C) Dedifferentiation is a pre-requisite for re-differentiation. (D) Abscisic acid is a plant growth promoter. (E) Apical dominance promotes the growth of lateral buds.
A, B and E only
B, D and E only
A, B and C only
A, C and D only
Which of the following hormones released from the pituitary is actually synthesized in the hypothalamus?
Follicle-stimulating hormone (FSH)
Adrenocorticotropic hormone (ACTH)
Luteinizing hormone (LH)
Anti-diuretic hormone (ADH)
Which of the following is an example of non-distilled alcoholic beverage produced by yeast?
Beer
Rum
Whisky
Brandy
What is the pattern of inheritance for polygenic trait?
Autosomal dominant pattern
X-linked recessive inheritance pattern
Mendelian inheritance pattern
Non-mendelian inheritance pattern
Match List-I with List-II.
A-I, B-IV, C-III, D-II
A-II, B-III, C-I, D-IV
A-I, B-II, C-IV, D-III
A-I, B-II, C-III, D-IV
Which of the following is an example of a zygomorphic flower?
Pea
Chilli
Petunia
Datura
Which of following organisms cannot fix nitrogen?
A. Azotobacter
B. Oscillatoria
C. Anabaena
D. Volvox
Which one of the following is an example of ex-situ conservation?
Zoos and botanical gardens
Protected areas
National Park
Wildlife Sanctuary
Who is known as the father of Ecology in India?
Ram Udar
Birbal Sahni
S.R. Kashyap
Ramdeo Misra
Given below are two statements : Statement I : In the RNA world, RNA is considered the first genetic material evolved to carry out essential life processes. RNA acts as a genetic material and also as a catalyst for some important biochemical reactions in living systems. Being reactive, RNA is unstable. Statement II : DNA evolved from RNA and is a more stable genetic material. Its double helical strands being complementary, resist changes by evolving repairing mechanism. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
Given below are two statements : Statement I : Transfer RNAs and ribosomal RNA do not interact with mRNA. Statement II : RNA interference (RNAi) takes place in all eukaryotic organisms as a method of cellular defence. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
Match List-I with List-II.
A-I, B-II, C-IV, D-III
A-II, B-III, C-I, D-IV
A-I, B-II, C-III, D-IV
A-I, B-IV, C-III, D-II
All living members of the class Cyclostomata are :
Symbiotic
Ectoparasite
Free living
Endoparasite
Streptokinase produced by bacterium Streptococcus is used for
Liver disease treatment
Removing clots from blood vessels
Curd production
Ethanol production
Role of the water vascular system in Echinoderms is: A. Respiration and Locomotion B. Excretion and Locomotion C. Capture and transport of food D. Digestion and Respiration E. Digestion and Excretion Choose the correct answer from the options given below:
B and C Only
B, D and E Only
A and B Only
A and C Only
Match List-I with List-II.
| List-I | List-II | ||
|---|---|---|---|
| A | Pteridophyte | I | Salvia |
| B | Bryophyte | II | Ginkgo |
| C | Angiosperm | III | Polytrichum |
| D | Gymnosperm | IV | Salvinia |
A-III, B-IV, C-I, D-II
A-IV, B-III, C-II, D-I
A-III, B-IV, C-II, D-I
A-IV, B-III, C-I, D-II
Which are correct: A. Computed tomography and magnetic resonance imaging detect cancers of internal organs. B. Chemotherapeutics drugs are used to kill non-cancerous cells. C. α-interferon activate the cancer patients’ immune system and helps in destroying the tumour. D. Chemotherapeutic drugs are biological response modifiers. E. In the case of leukaemia blood cell counts are decreased.
A, B, C
A, C, D
A, C, E
B, D, E
What are the potential drawbacks in adoption of the IVF method?
High fatality risk to mother
Expensive instruments and reagents
Husband/wife necessary for being donors
Less adoption of orphans
Consider the following : A. The reductive division for the human female gametogenesis starts earlier than that of the male gametogenesis. B. The gap between the first meiotic division and the second meiotic division is much shorter for males compared to females. C. The first polar body is associated with the formation of the primary oocyte. D. Luteinizing Hormone (LH) surge leads to disintegration of the endometrium and onset of menstrual bleeding. Choose the correct answer from the options given below:
B and D are true
B and C are true
A and B are true
A and C are true
In bryophytes, the gemmae help in which one of the following?
Nutrient absorption
Gaseous exchange
Sexual reproduction
Asexual reproduction
Given below are two statements: one is labelled as Assertion (A), and the other is labelled as Reason (R). Assertion (A): The primary function of the Golgi apparatus is to package the materials made by the endoplasmic reticulum and deliver it to intracellular targets and outside the cell. Reason (R): Vesicles containing materials made by the endoplasmic reticulum fuse with the cis face of the Golgi apparatus, and they are modified and released from the trans face of the Golgi apparatus. In the light of the above statements, choose the correct answer from the options given below:
A is true but R is false
A is false but R is true
Both A and R are true and R is the correct explanation of A
Both A and R are true but R is not the correct explanation of A
Which one of the following statements refers to Reductionist Biology?
Chemical approach to study and understand living organisms
Behavioural approach to study and understand living organisms
Physico-chemical approach to study and understand living organisms
Physiological approach to study and understand living organisms
After maturation, in primary lymphoid organs, the lymphocytes migrate for interaction with antigens to secondary lymphoid organ(s) / tissue(s) like
thymus
bone marrow
spleen
lymph nodes
Match List I with List II:
| List-I | List-II |
| A. The Evil Quartet | I. Cryopreservation |
| B. Ex situ conservation | II. Alien species invasion |
| C. Lantana camara | III. Causes of biodiversity losses |
| D. Dodo | IV. Extinction |
Choose the option with all correct matches.
A-III, B-IV, C-II, D-I
A-III, B-II, C-IV, D-I
A-III, B-II, C-I, D-IV
A-III, B-I, C-II, D-IV
How many meiotic and mitotic divisions need to occur for the development of a mature female gametophyte from the megaspore mother cell in an angiosperm plant?
1 Meiosis and 3 Mitosis
No Meiosis and 2 Mitosis
2 Meiosis and 3 Mitosis
1 Meiosis and 2 Mitosis
Which of the following type of immunity is present at the time of birth and is a non-specific type of defence in the human body?
Cell-mediated Immunity
Humoral Immunity
Acquired Immunity
Innate Immunity
Given below are two statements : Statement I : Fig fruit is a non-vegetarian fruit as it has enclosed fig wasps in it. Statement II : Fig wasp and fig tree exhibit mutual relationship as fig wasp completes its life cycle in fig fruit and fig fruit gets pollinated by fig wasp. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
Given below are two statements: One is labelled as Assertion (A) and other is labelled as Reason (R). Assertion (A): Cells of the tapetum possess dense cytoplasm and generally have more than one nucleus. Reason (R): Presence of more than one nucleus in the tapetum increases the efficiency of nourishing the developing microspore mother cells. In light of the above statements, choose the most appropriate answer from the options given below:
A is true but R is false
A is false but R is true
Both A and R are true and R is the correct explanation of A
Both A and R are true but R is NOT the correct explanation of A
From the statements given below choose the correct option : A. The eukaryotic ribosomes are 80S and prokaryotic ribosomes are 70S. B. Each ribosome has two sub-units. C. The two sub-units of 80S ribosome are 60S and 40S while that of 70S are 50S and 30S. D. The two sub-units of 80S ribosome are 60S and 20S and that of 70S are 50S and 20S. E. The two sub-units of 80S are 60S and 30S and that of 70S are 50S and 30S.
A, B, E are true
B, D, E are true
A, B, C are true
A, B, D are true
Which one of the following enzymes contains 'Haem' as the prosthetic group?
Succinate dehydrogenase
Catalase
RuBisCo
Carbonic anhydrase
What is the name of the blood vessel that carries deoxygenated blood from the body to the heart in a frog?
Pulmonary vein
Vena cava
Aorta
Pulmonary artery
Given below are the stages in the life cycle of pteridophytes. Arrange the following stages in the correct sequence. A. Prothallus stage B. Meiosis in spore mother cells C. Fertilisation D. Formation of archegonia and antheridia in gametophyte. E. Transfer of antherozoids to the archegonia in presence of water. Choose the correct answer from the options given below:
D, E, C, A, B
E, D, C, B, A
B, A, D, E, C
B, A, E, C, D
The blue and white selectable markers have been developed which differentiate recombinant colonies from non-recombinant colonies on the basis of their ability to produce colour in the presence of a chromogenic substrate. Given below are two statements about this method: Statement I : The blue coloured colonies have DNA insert in the plasmid and they are identified as recombinant colonies. Statement II : The colonies without blue colour have DNA insert in the plasmid and are identified as recombinant colonies. In the light of the above statements, choose the most appropriate answer from the options given below:
Statement I is correct but Statement II is incorrect
Statement I is incorrect but Statement II is correct
Both Statement I and Statement II are correct
Both Statement I and Statement II are incorrect
Which of the following microbes is NOT involved in the preparation of household products?
A. Aspergillus niger
B. Lactobacillus
C. Trichoderma polysporum
D. Saccharomyces cerevisiae
Silencing of specific mRNA is possible via RNAi because of
Complementary tRNA
Non-complementary ssRNA
Complementary dsRNA
Inhibitory ssRNA
The complex II of mitochondrial electron transport chain is also known as
Cytochrome c oxidase
NADH dehydrogenase
Cytochrome
Succinate dehydrogenase
While trying to find out the characteristic of a newly found animal, a researcher did the histology of adult animal and observed a cavity with presence of mesodermal tissue towards the body wall but no mesodermal tissue was observed towards the alimentary canal. What could be the possible coelome of that animal?
Schizocoelomate
Spongocoelomate
Acoelomate
Pseudocoelomate
Given below are two statements: Statement I: In a floral formula ⊕ stands for zygomorphic nature of the flower, and G stands for inferior ovary. Statement II: In a floral formula ⊕ stands for actinomorphic nature of the flower and G stands for superior ovary. In the light of the above statements, choose the correct answer from the options given below:
Statement I is correct but Statement II is incorrect
Statement I is incorrect but Statement II is correct
Both Statement I and Statement II are correct
Both Statement I and Statement II are incorrect
Given below are two statements: Statement I: In ecosystem, there is unidirectional flow of energy of sun from producers to consumers. Statement II: Ecosystems are exempted from 2nd law of thermodynamics. In the light of the above statements, choose the most appropriate answer from the options given below:
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
Which of the following is the unit of productivity of an Ecosystem?
With the help of given pedigree, find out the probability for the birth of a child having no disease and being a carrier (has the disease mutation in one allele of the gene) in F₃ generation.
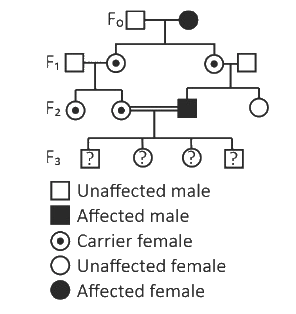
1/8
Zero
1/4
1/2
In the seeds of cereals, the outer covering of endosperm separates the embryo by a protein-rich layer called :
Integument
Aleurone layer
Coleoptile
Coleorhiza
Match List I with List II:
| List-I | List-II |
| A. Chlorophyll a | (I) Yellow-green |
| B. Chlorophyll b | (II) Yellow |
| C. Xanthophylls | (III) Blue-green |
| D. Carotenoids | (IV) Yellow to Yellow-orange |
A-I, B-II, C-IV, D-III
A-I, B-IV, C-III, D-II
A-III, B-IV, C-II, D-I
A-III, B-I, C-II, D-IV
Who proposed that the genetic code for amino acids should be made up of three nucleotides?
Jacque Monod
Franklin Stahl
George Gamow
Francis Crick
Histones are enriched with -
Phenylalanine & Leucine
Phenylalanine & Arginine
Lysine & Arginine
Leucine & Lysine
Which of the following enzyme(s) are NOT essential for gene cloning?
C and D only
A and B only
D and E only
B and C only
A specialised membranous structure in a prokaryotic cell which helps in cell wall formation, DNA replication and respiration is
Cristae
Endoplasmic Reticulum
Mesosome
Chromatophores
Which factor is important for termination of transcription?
(rho)
(gamma)
(alpha)
(sigma)
Which of the following statement is correct about location of the male frog copulatory pad?
Second digit of fore limb
First digit of the fore limb
First and Second digit of fore limb
First digit of hind limb
Which of the following diagrams is correct with regard to the proximal (P) and distal (D) tubule of the Nephron.
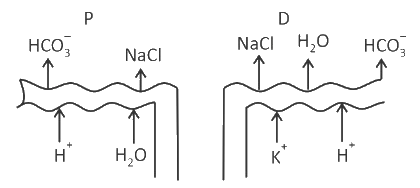
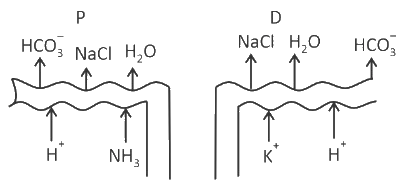
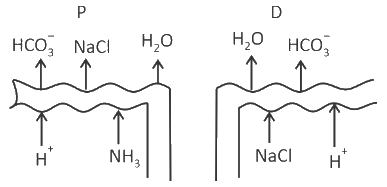
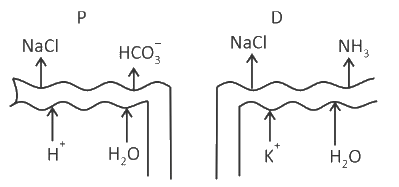
Identify the statement that is NOT correct.
Antigen binding site is located at C-terminal region of antibody molecules.
Constant region of heavy and light chains are located at C-terminus of antibody molecules
Each antibody has two light and two heavy chains.
The heavy and light chains are held together by disulfide bonds.
Match List I with List II:
| List I | List II | ||
|---|---|---|---|
| A. | Scutellum | I. | Persistent nucellus |
| B. | Non-albuminous seed | II. | Cotyledon of Monocot seed |
| C. | Epiblast | III. | Groundnut |
| D. | Perisperm | IV. | Rudimentary cotyledon |
Choose the option with all correct matches.
A-IV, B-III, C-I, D-II
A-II, B-IV, C-III, D-I
A-II, B-III, C-IV, D-I
A-IV, B-III, C-II, D-I
Find the statement that is NOT correct with regard to the structure of monocot stem.
Vascular bundles are conjoint and closed.
Phloem parenchyma is absent.
Hypodermis is parenchymatous.
Vascular bundles are scattered.
Twins are born to a family that lives next door to you. The twins are a boy and a girl. Which of the following must be true?
They were conceived through in vitro fertilization.
They have 75% identical genetic content.
They are monozygotic twins.
They are fraternal twins.
Sweet potato and potato represent a certain type of evolution. Select the correct combination of terms to explain the evolution.
Homology, convergent
Analogy, divergent
Analogy, convergent
Homology, divergent
Which one of the following phytohormones promotes nutrient mobilization which helps in the delay of leaf senescence in plants?
Gibberellin
Cytokinin
Ethylene
Abscisic acid
Why can't insulin be given orally to diabetic patients?
Because of structural variation
Its bioavailability will be increased
Human body will elicit strong immune response
It will be digested in Gastro-Intestinal (GI) tract
Name the class of enzyme that usually catalyze the following reaction : Where, a group other than hydrogen a substrate another substrate
Transferase
Ligase
Hydrolase
Lyase
Given below are two statements : Statement I : The DNA fragments extracted from gel electrophoresis can be used in construction of recombinant DNA. Statement II : Smaller size DNA fragments are observed near anode while larger fragments are found near the wells in an agarose gel. In the light of the above statements, choose the most appropriate answer from the options given below :
Statement I is correct but statement II is incorrect
Statement I is incorrect but statement II is correct
Both statement I and statement II are correct
Both statement I and statement II are incorrect
The correct sequence of events in the life cycle of bryophytes is A. Fusion of antherozoid with egg. B. Attachment of gametophyte to substratum. C. Reduction division to produce haploid spores. D. Formation of sporophyte. E. Release of antherozoids into water. Choose the correct answer from the options given below :
B, E, A, D, C
D, E, A, B, C
D, E, A, C, B
B, E, A, C, D
Genes R and Y follow independent assortment. If RRYy produce round yellow seeds and rryy produce wrinkled green seeds, what will be the phenotypic ratio of the F2 generation?
Phenotypic ratio - 9 : 3 : 3 : 1
Phenotypic ratio - 9 : 7
Phenotypic ratio - 1 : 2 : 1
Phenotypic ratio - 3 : 1
Each of the following characteristics represent a Kingdom proposed by Whittaker. Arrange the following in increasing order of complexity of body organization. A. Multicellular heterotrophs with cell wall made of chitin. B. Heterotrophs with tissue/organ/organ system level of body organization. C. Prokaryotes with cell wall made of polysaccharides and amino acids. D. Eukaryotic autotrophs with tissue/organ level of body organization. E. Eukaryotes with cellular body organization. Choose the correct answer from the options given below :
A, C, E, D, B
C, E, A, B, D
A, C, E, B, D
C, E, A, D, B
Match List-I with List-II.
| List-I | List-II | ||
|---|---|---|---|
| A | Centromere | I | Mitochondrion |
| B | Cilium | II | Cell movement |
| C | Cristae | III | Cell division |
| D | Cell membrane | IV | Phospholipid Bilayer |
A-IV, B-II, C-III, D-I
A-II, B-III, C-I, D-IV
A-I, B-II, C-III, D-IV
A-II, B-I, C-IV, D-III
Which one of the following equations represents the Verhulst-Pearl Logistic Growth of population?
Match List-I with List-II.
| List-I | List-II |
| A. Emphysema | II. Damaged alveolar walls and decreased respiratory surface |
| B. Angina Pectoris | III. Acute chest pain when not enough oxygen is reaching to heart muscle |
| C. Glomerulonephritis | IV. Inflammation of glomeruli of kidney |
| D. Tetany | I. Rapid spasms in muscle due to low Ca²⁺ in body fluid |
A-II, B-IV, C-III, D-I
A-II, B-III, C-IV, D-I
A-III, B-I, C-IV, D-II
A-III, B-I, C-II, D-IV
Cardiac activities of the heart are regulated by: A. Nodal tissue B. A special neural centre in the medulla oblongata C. Adrenal medullary hormones D. Adrenal cortical hormones Choose the correct answer from the options given below :
A, C and D Only
A, B and D Only
A, B and C Only
A, B, C and D
Drill the chapters tested in NEET 2025
Note the chapters where you slipped up in this paper, then revise via NCERT-aligned notes, interactive widgets, and chapter-wise mock tests for all four subjects.
Physics
28 NEET chapters
Chemistry
28 NEET chapters
Botany
18 NEET chapters
Run NEET 2025 as a real exam.
Source: National Testing Agency (NTA), NEET-UG 2025 Question Paper, Code 48. Solutions written for educational use, NCERT-aligned. Verify your answers against the official NTA answer key.